|
12/25/2023 0 Comments Signal transduction pathway examples 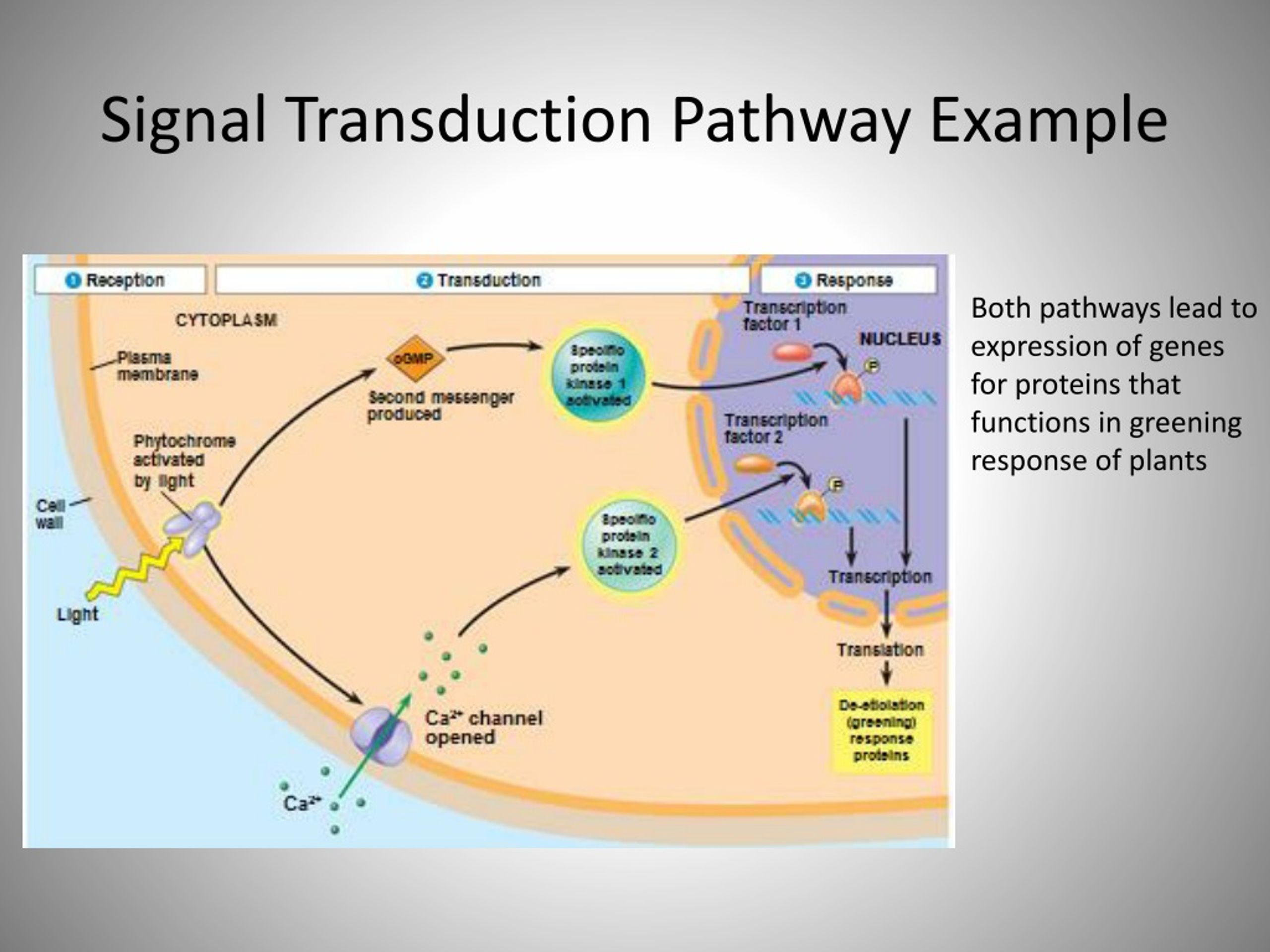
One of the well-known targets of PKA is CRE-binding protein (CREB) PKA phosphorylates CREB on a specific serine residue. The regulatory subunits of PKA (also known as A-kinase) are important for the sub-cellular localization of PKA, which is facilitated by the interaction of A-kinase anchoring proteins (AKAPs) with the regulatory subunits. The binding of cyclic AMP to the regulatory subunits induces a specific conformational change and results in the dissociation of the complex, which leads to the activation of the catalytic subunits. In the inactive state, PKA is composed of a complex of two catalytic subunits and two regulatory subunits. Once activated, PKA can phosphorylate many proteins on serine/threonine sites. In mammalian cells, GPCR-induced cyclic AMP results in the activation of the cyclic AMP-dependent protein kinase (PKA). Pertussis toxin that causes pertussis (whooping cough) also can hydrolyze ADP-ribosylation of the α subunit of Gi, which inhibits its binding with GPCR, which results in the α subunit to remain in the GDP-bound state and makes it unable to regulate downstream signaling events. Sustained and elevated concentration of cycle AMP in intestinal epithelial cells results in diarrhea. Cholera toxin interferes with the activity of the α subunit by ADP-ribosylation, causing it to remain active. Both Gs and Gi are targets of bacterial toxins. However, binding an inhibitory G protein (Gi) to a GPCR can inhibit cyclic AMP synthesis. GPCRs that stimulate the production of cyclic AMP are often coupled to the stimulatory G protein (Gs), which activates adenylyl cyclase and increases cyclic AMP levels. GPCRs activate various intracellular signaling, including generating second messengers such as cyclic AMP and inositol phospholipids. There are currently 25 known GAPs in the human genome. RGS proteins function as subunit-specific GTPase activating proteins (GAPs). The GTPase activity of the α subunit of G protein is significantly enhanced when it interacts with a specific regulator of G protein signaling (RGS). The α subunit of G protein has a GTPase activity, and once it hydrolyzes GTP to GDP, it becomes inactive. The exchange of GDP with GTP results in a conformational change in the G protein, which leads to its activation. By doing so, GPCR acts like a guanine nucleotide exchange factor (GEF). However, when a GPCR is activated, it induces the α subunit to release GDP and instead binds to guanosine triphosphate (GTP). When it is inactive, the α subunit of the G protein is bound to guanosine diphosphate (GDP). G proteins have three subunits (α,β, and γ). In some cases, G proteins interact with the GPCR before the receptor is activated in other instances, G protein interacts with GPCR only after stimulation with a ligand. The G protein is associated with the plasma membrane at the cytoplasmic side, connecting the GPCR to either enzymes or ion channels. The binding of extracellular ligands initiates the signal transduction cascade by triggering conformational changes in the receptor that promote heterotrimeric GTP-binding protein (G protein) activation. GPCRs were named for their common ability to associate with heterotrimeric G proteins (Gαβγ). G protein-coupled receptors (GPCRs) are integral membrane proteins that form the fourth largest superfamily in the human genome. Signal transduction through G proteins is the most prominent feature of GPCRs, initiated by a ligand-GPCR interaction at the cell surface level. Targeting of GPCRs is hence widely utilized for therapeutic intervention GPCRs correspond to 30% of all identified drug targets and remain major targets for new drug development. Impaired ligand concentration, GPCR protein expression, or mutation and signaling are implicated in many pathophysiological conditions, including central nervous system (CNS) disorders, cardiovascular and metabolic diseases, respiratory malfunctions, gastrointestinal disorders, immune diseases, cancer, musculoskeletal pathologies, and eye diseases. The GPCR superfamily comprises at least five structurally distinct subfamilies: Glutamate, Rhodopsin, Adhesion, Frizzled/Taste2, and Secretin receptor families. About 90% of all GPCRs belong to the rhodopsin family. The human genome encodes nearly 800 GPCRs, representing over 3% of human genes. GPCRs recognize a wide variety of signals ranging from photons to ions, proteins, neurotransmitters, and hormones. G protein-coupled receptors (GPCRs) are integral membrane proteins containing an extracellular amino terminus, seven transmembrane α-helical domains, and an intracellular carboxy terminus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
